Medical Devices: Increase innovation through multiphysics-CFD Design Exploration
Innovation through CFD and Design Exploration for medical devices
The rapidly increasing cost of clinical research has been a key obstacle to innovation in the development of new medical devices, which is bad news for patients, manufacturers, regulatory authorities and the sector as a whole. To a greater extent, simulation-based product development and virtual prototyping are being recognized by the FDA as valid tools to supplement experimental testing and clinical trials and accelerate regulatory approval. Initiatives like the ASME V&V 40, MDIC, or Avicenna, drive and guide these efforts and lobby for the increased usage of simulation for the support of benchtop and clinical trials in the medical device industry.
However, only deploying simulation in a “validation and verification” context is missing a large part of its potential value in the product development process. Using Simcenter STAR-CCM+ allows engineers to turn every simulation of a medical device into a design exploration study, in which results of multiphysics-CFD simulations automatically drive parametric changes in the design of a device.
Used in this manner, simulation enables medical device companies to perform investigations where experimentation does not exist, or is costly or unethical, reducing dependence on animal models and human data. While clinical trials can realistically capture data from a few hundred patients in a trial, design exploration studies allow engineers to automatically simulate thousands of design variations and a full range of usage scenarios. This increases confidence in the devices, increases innovation and reduces the cost of clinical trials.
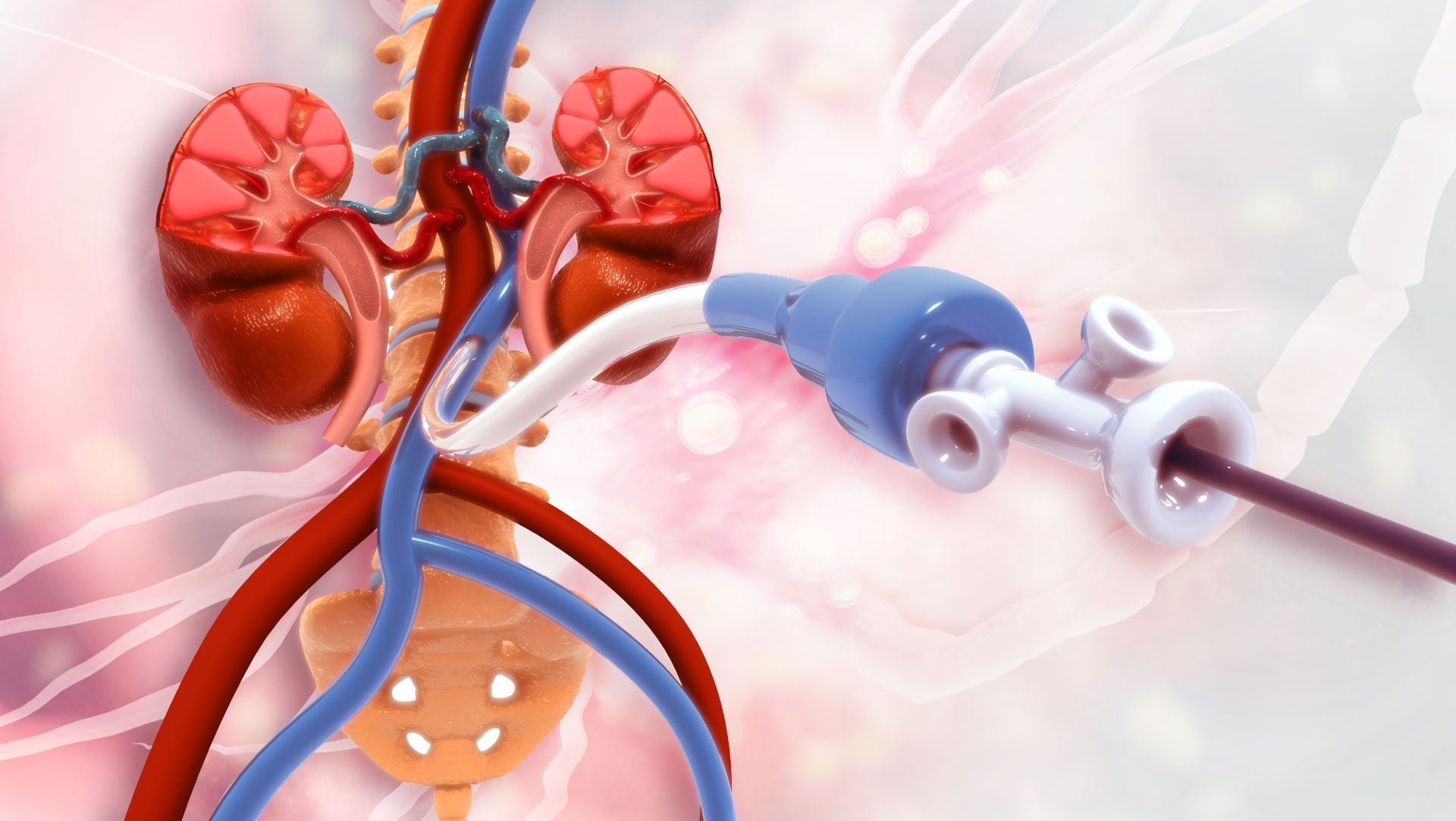
This comprehensive webinar will quantify the benefits of deploying design exploration in the development of medical devices, through the example of a dialysis catheter simulation.
Presenters:
- Thomas McIlwain: University of Washington, Neural Engineering
- Aaron Godfrey: Siemens PLM Software