Successful growth with end-to-end innovation platform
Sensile Medical achieves solid growth with Siemens Digital Industries Software solutions and Quadrix
Sensile Medical
Sensile Medical is an internationally leading Swiss medical technology company that develops and manufactures high-tech micropumps for the medical sector.
http://www.sensile-medical.com- Headquarters:
- Haegendorf, Switzerland
- Products:
- NX, NX CAM, Simcenter Products, Femap, Solid Edge, Teamcenter
- Industry Sector:
- Medical devices & pharmaceuticals
I was used to the application of another CAD system, but was immediately able to find my way through the intuitive user guidance of Solid Edge.
Sensile Medical AG
Innovative medical devices
Sensile Medical AG, a medical devices company based in Haegendorf, Switzerland, develops and produces innovative rotary pumps for highly precise dosing and application of liquids. Starting from the first development, the company has used Solid Edge® software from product lifecycle management (PLM) specialist Siemens Digital Industries Software for 3D computer-aided design (CAD). Later it was supplemented by Simcenter Femap™ software for simulation and NX™ software for computer-aided manufacturing (CAM) for numerical control (NC) programming.
Using the industry-leading Teamcenter® software for product data management (PDM), the company is heading for future success: the Siemens Digital Industries Software applications empower strong growth and compliance to various international requirements and standards of the industry.
When the company was founded, it focused on the SenseCore technology for precise drug delivery: the core element consists of only two micromechanical plastic parts – a housing and rotary pump shaft. ”The main benefit is simplicity,“ explains Daniel Grossenbacher, head of mechanical engineering at Sensile Medical. ”Without any additional valves, the pump sucks the fluid in one stroke and injects it during the same rotation.“
When the company was evaluating CAD systems, many of the solutions failed to model the complex geometry of the housing. ”Comprehensive features for surface creation were an important criterion when selecting Solid Edge,“ recalls Gerhard Eimer, managing director of Quadrix AG in Flawil, Switzerland, a market-leading value-added reseller for Siemens Digital Industries Software.
A startup company, Sensile Medical holds a family of more than 30 patents concerning this technology. The small, compact injection-molded parts require manufacturing accuracy within hundredths of millimeters. Today, a broad range of innovative products has evolved from the core technology.

Bridge between industry and patient
Sensile Medical is developing task-specific solutions in the areas of pharmaceutical and biotech for customers that want to deliver fluid medicine to the bodies of patients. In 2015, the company was nominated as ”Innovation Leader” with the Swiss Technology Award, the major distinction for innovation and technology transfer in Switzerland. ”Our devices are easy to use and safe in application,” says Derek Brandt, CEO of Sensile Medical. ”The patients benefit from that and a better therapy at lower costs.”
Sensile Medical’s small volume wearable devices are intended for home use.
The first area of application is heart insufficiency, and others are in evaluation. The global pharmaceutical and life sciences industries are developing new liquid medicine and active ingredients, eager to find easy and safe modes of delivery. This pressure to grow led the company to double the number of employees in the last three years to 60 people in 2016. They work in engineering, electrical technology, software development and microsystems technology.
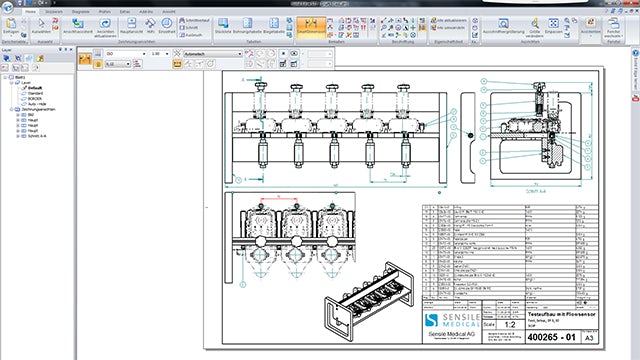
Broad development platform
Currently, two industrial designers and eight mechanical engineers are using Solid Edge. ”I was used to the application of another CAD system, but was immediately able to find my way through the intuitive user interface of Solid Edge,” says Grossenbacher. ”Starting on my own, I was surprised by the broad range and depth of features, especially when creating surfaces and defining freeform surfaces.”
When creating designs from customer-specific requirements, creative departments that are guided by custom specifications, safety and ergonomics in creation are at an advantage. In the preliminary project phase, the initial visualizations should first and foremost convey the product vision. Later in the process, customers want to have mock-ups created using rapid prototyping processes in their hands right away. The mock-ups are then checked in usability studies for size, feel and ergonomics. The most exacting aesthetic requirements are in place from the initial draft to production on injection molding machines. “In addition, having a system with distinct options for freeform surfaces really comes in handy,” says Grossenbacher.
For a production-ready design, fillets and draft angles need to be considered. A high surface quality should not be disrupted by visible sprue marks or indentations at the injection site. In the process, simulation with the Simcenter Femap software program performs calculations in accordance with the finite element method (FEM) and is directly integrated with Solid Edge. “These quick and easy simulations enable us to save time in the development process,” says Grossenbacher. “We gather valuable experiences in order to expedite our development, even though the supplier carries out his own filling simulations in the tool design. ”With associative links between the simulation and development model, the two Siemens Digital Industries Software solutions accelerate change cycles.
Professional FEM simulation
Simcenter Femap became a part of the integrated development platform at a very early stage. After the Swiss Federal Institute of Technology Zurich had tested the first prototypes of the SenseCore pump, Sensile Medical was able to evaluate three integrated simulation solutions. “The Simcenter Femap simulation returned a pump pressure that very closely matched the pump pressure determined on the prototypes,” Eimer remembers. “The closest competitor was at 70 percent.” Today, along with filling simulation, Simcenter Femap is primarily used to perform stress analyses. On the one hand, stress analyses provide proof to clients and authorities that the complex devices are able to withstand the anticipated loads. On the other hand, they are an important resource due to the pressure of miniaturization. “Stress analyses give us the assurance that, for example, wall thickness ratios do not cause any problems in production,” reports Grossenbacher. “Thanks to the assurance from the stress analyses, we often save the critical percentages that would otherwise enlarge a product.”
The NX CAM system, directly linked to Solid Edge, affords a direct route to the workshop for the creation of NC programs. However, outputting STL files for rapid prototyping methods has become more important. “This provides us with finished prototypes within three days,” says Grossenbacher.
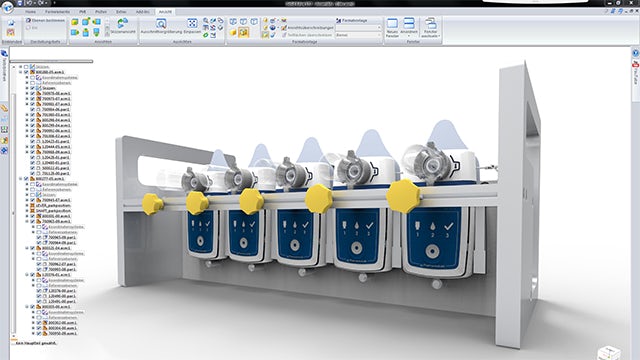
Flexible assembly design
Changes to product designs are made easier by associative linking, as well as by adaptive design in the assembly – a feature of Solid Edge that is used often at Sensile Medical. It extends all the way to the bill of material: when using three pieces of the same cable that have different lengths, designers aren’t required to enter it three times due to the different shapes. Motion analyses are also performed in the assemblies with the utilities of the CAD system. Grossenbacher is particularly impressed by the collision detection: “I didn’t know that it was possible to perform design analyses so easily and directly in the basic version of the CAD system,” he says. “You simply select a component and have all the collisions displayed in color. Once the correct tick mark has been placed in the menu, the system gives a warning about resulting collisions for each change.”
Built-in data management
Another special feature of Solid Edge is the built-in function of a secure data management program. With every access the status of components and documents is updated, so that simultaneous access by multiple users is impossible. Components and documents can be blocked or released; attributes like material, user or supplier are maintained with the components – in short, built-in functions for product data management ensure a structured approach and data security. “Without these functions, cooperation among eight engineers would have been impossible,” reports Grossenbacher. “This basic functionality has enabled us to master data management in accordance with the regulatory bodies for medical technology.”
Teamcenter – investment in the future
Sensile Medical must also comply with the stringent requirements of industry standards, including ISO 13485 and those of FDA/cGMP (21CFR 820) for medical devices. The application of Usability Engineering according to IEC 62366 for medical devices and Risk Management in accordance with ISO 14971 must likewise be observed.
Due to continuous growth, new projects will require many more employees. Therefore Sensile Medical has already teamed with Siemens Digital Industries Software to implement Teamcenter, the world’s leading PDM/PLM solution. Teamcenter runs in the background whenever users are working with Solid Edge and can be accessed from the Solid Edge user interface. “We are banking on Teamcenter due to its expandability,” says Grossenbacher. “In medical technology, a powerful PDM system can be a determining factor in success.”
With Teamcenter fully implemented, all user groups including industrial designers, project managers and designers are able to access all data and extract information.
Customizable workflows enforce change and release processes, and user authorizations and user-specific views simplify the overview. “Everything works the way we envisioned,” says Grossenbacher.
Now the company is primed for the next growth step. “We plan to hire 30 additional employees this year,” says Brandt. “Currently we are struggling to find additional engineers.”

Development platform from a single source
The company can access services and support, including those of Quadrix as a contact for the product development team at Siemens Digital Industries Software. “Our long-term partnership has paid off,” says Brandt. The company appreciates the expert consultation for future expansion phases, support in installation of pre-tested, regular updates, the comprehensive internal and external training program and the support hotline. “At Quadrix, we are always able to reach a specialist who can provide us with information in a professional manner,” says Grossenbacher. “We are never on hold, the way it was with the other vendors.”
We are banking on Teamcenter due to its expandability. In medical technology, a powerful PDM system can be a determining factor in success.
Sensile Medical AG
