Driving competitive advantage through PLM
NX and Teamcenter help Wright achieve innovation, speed and compliance goals
Wright Medical Technology
Wright Medical Technology, Inc. is a global orthopedic medical device company specializing in the design, manufacture and marketing of reconstructive joint devices and biologics. Wright’s product offerings include large joint implants for the hip and knee; extremity implants for the hand, elbow, shoulder, foot and ankle; and both synthetic and tissue-based bone graft substitute materials. Wright’s common stock is listed on the Nasdaq Global Select Market under the symbol “WMGI.”
https://www.wright.com/- Headquarters:
- Arlington, United States
- Products:
- NX, Teamcenter
- Industry Sector:
- Medical devices & pharmaceuticals
NX is the most powerful design software I’ve ever used.
Wright Medical Technology
Commitment to medical breakthroughs in orthopedics
Wright Medical Technology, Inc. (Wright) is a global orthopedic medical device company specializing in the design, manufacture and marketing of reconstructive joint devices and biologics. Wright’s product offerings include large joint implants for the hip and knee; extremity implants for the hand, elbow, shoulder, foot and ankle; and both synthetic and tissue-based bone graft substitute materials. Headquartered in Arlington, Tennessee, USA, the company has been in business for more than 50 years, employs approximately 1,000 and is listed on the Nasdaq Global Select Market under the symbol “WMGI.”
With a commitment to evolving medical breakthroughs in the field of orthopedics, Wright management saw an opportunity to think bigger and push the envelope of innovation, quality and compliance by investing in product lifecycle management (PLM) technology – NX™ software and Teamcenter® platform – from Siemens Digital Industries Software.
Wright uses Siemens’ solutions for product development, data and medical records management, collaboration, compliance, traceability and reporting, including using the software to meet FDA regulatory requirements.
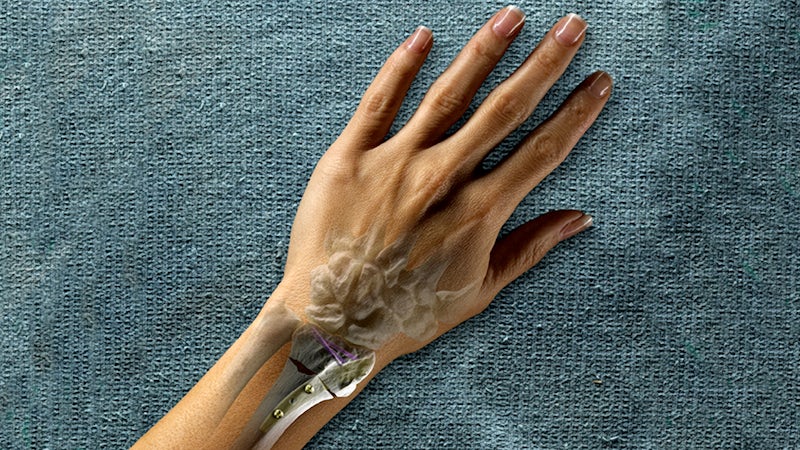
An example of Wright’s applying PLM for innovation purposes is demonstrated in its use of NX. “The distal radius is the forearm bone on the thumb side and it’s the most commonly fractured bone,” says Penny Rasmussen, marketing manager for the MICRONAIL® project. “Wright had a goal to make a less invasive, less painful way to repair this common fracture. And that solution was the MICRONAIL® Intramedullary Fixation Implant. NX helped Wright bring that product to the market in record time.”
Visualization, collaboration
According to Chad Schwartz, lead design engineer for the MICRONAIL® project, the assembly package for NX enables Wright to use the implant to design instruments for ease of development and change. “As far as different sizes for implants or something the doctor wants changed, it’s easy to do because everything updates succinctly.” He notes, “NX is the most powerful design software I’ve ever used. The solid models, the assemblies, all of the things that come together in the modeling aspect produce better results. You can see things fit together in a way that you can’t do in other software. You see your finished product before it’s actually a finished product.”
Teamcenter extends the company’s ability to flourish. “Teamcenter allows Wright to be more competitive in the marketplace by allowing manufacturing, engineering, quality and marketing all to work in parallel with one another,” says Schwartz. “That means saving time, getting our product on the market in a faster manner and beating the competition more often than not. Teamcenter is probably the best thing that’s happened to Wright, as far as bringing everything together and making it all one. The time savings alone are worth it. Instead of having to go and find the right design data file in the file cabinet, you have everything right there at your fingertips.”
While data re-use is important, collaboration is critical. Schwartz points out, “Before Teamcenter, we had to send the doctor a rapid prototype model. The doctor would send it back to us with markings and tell us how to change it. Now the doctor can look at our model as we rotate it or maybe an animation that we set up in Teamcenter. We get quicker response. Instead of maybe a three-day turnaround, we’ve got it down to a couple of hours.” In addition, he notes, “Our distributors throughout the country can log on and look at what we’re designing right then. Show a doctor what’s coming. And they can decide right then whether or not they’d like to use it.”
As new products are developed and manufactured, Wright has expanded the use of Teamcenter to manage all kinds of engineering specifications in a single platform. This includes gradually achieving the company’s objective of an electronic Device Medical Record (DMR).
Compliance
Complying with FDA regulations represents another important area where an advantage gained can be significant in terms of turnaround, especially as regulations are prone to change often in the medical industry. In fact, Wright has dramatically enhanced the availability of compliance data and reduced the time it takes to access that data. Previously a process that was paperwork intensive – sometimes taking days, weeks or even months – device history is now immediately available. Using Teamcenter, Wright can access the information with a few clicks.
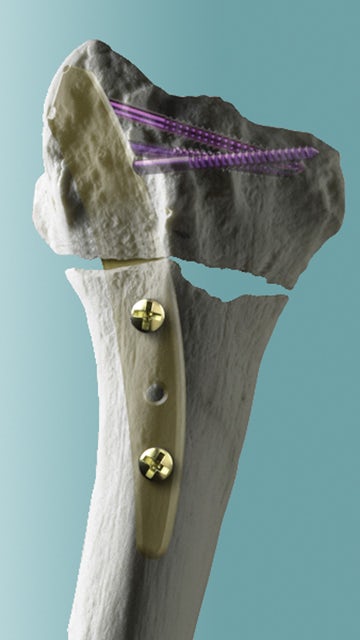
Performance
Wright moved to a product-centric system not just for reasons of innovation, speed and compliance on the front end of the process, but because products need to last the lifecycle of their patients, which can be decades.
The company’s approach to orthopedic medical device delivery – strategically, operationally and technologically – is proving to be quite effective.
Moreover, Wright’s financial performance is strong. Rasmussen notes, “We’re growing. We’re increasing our market share every year. It has a lot to do with the systems that we run, Teamcenter being the main one.”
We’re increasing our market share every year. It has a lot to do with the systems that we run, Teamcenter being the main one.
Wright Medical Technology