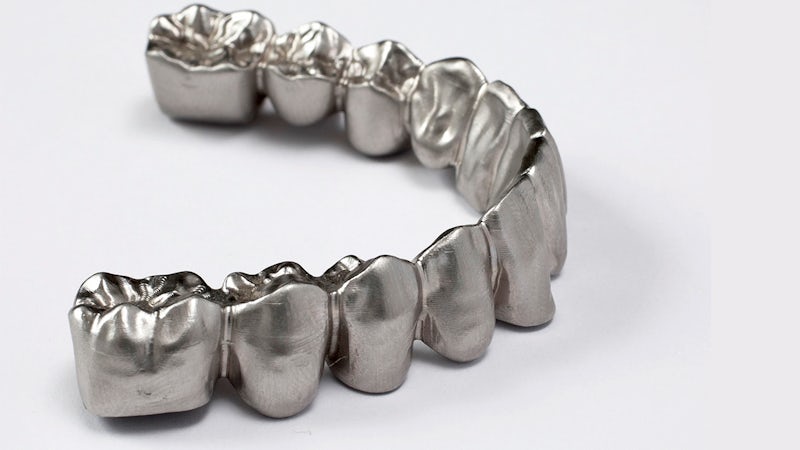
With Solid Edge and Simcenter Femap, dental implant manufacturer opens up new markets
Using Siemens Digital Industries Software solutions helps Biotec launch new brand and image
Biotec
Under the BTK brand, Biotec manufactures and sells dental implants. The company performs research, innovation and clinical studies, managing the entire process for the design and production of certified and safe medical devices.
http://www.bioteconline.com- Headquarters:
- Povolaro di Dueville, Vicenza, Italy
- Products:
- Simcenter Products, Femap, Solid Edge
- Industry Sector:
- Medical devices & pharmaceuticals
We selected Solid Edge because it is powerful, intuitive and fast. Many design tools require long preparation and setup, whereas we often design entire product families, so we need a very dynamic and flexible approach. We must have all the features of parametric CAD in intuitive and easyto-use software. Solid Edge fits that description.
Biotec
The Smile System
Biotec Srl was established in 1998 as a subcontractor for the production of medical devices. Based in Povolaro di Dueville, near Vicenza, Italy, the company decided to invest in the development of an advanced dental implant program under its own brand.
“From a strategic standpoint, we realized that subcontracting was not a long-term vision, so we gathered the entire production under the Biotec brand and, in 2009, we undertook a new project to relaunch our business, involving all areas from technology to marketing,” says Dr. Andrea Peloso, managing director of Biotec. “That’s how the ‘btk’ brand was born, propelling Biotec into the business of cosmetic surgery and dental implants. With its three letters, the new brand maintains an association with the company’s name and, at the same time, sums up its core values: biocompatibility, technology and know-how.”
Fittingly, the company’s tagline is “the smile system™.”
Biotec employs about 40 people, and has doubled its headcount in the last three years. Sixty percent of the business comes from Italy and the rest is generated from Southern Europe, the Middle East, North Africa and Eastern Europe. New markets are opening at regular intervals from Libya to Russia, often requiring long certification procedures before products can be marketed. In some cases, the waiting period can be up to two years.


“Powerful, intuitive and fast”
A dental implant is not a simple product: It has small dimensions, needs to perform multiple functions and is used by demanding doctors.
“In recent years, doctors have shown increasing interest in design,” says Peloso. “They know about technology and ask to be involved. It’s great to have their help developing high-tech and innovative products provided that we have tools that enable us to design concurrently with physicians, showing them the conceptual prototype of an implant as well as the final product.”
To meet these new requirements, Biotec management decided to adopt product lifecycle management (PLM) technology in 2010, and chose Solid Edge® software from Siemens Digital Industries Software. Solid Edge provides advanced computeraided design (CAD) technology for accelerated product development, faster revisions and better data re-use.
“We selected Solid Edge because it is powerful, intuitive and fast,” says Igor Piccoli, research and development (R&D) manager at Biotec. “Many design tools require long preparation and setup, whereas we often design entire product families, so we need a very dynamic and flexible approach. We must have all the features of parametric CAD in intuitive and easy-to-use software. Solid Edge fits that description.”

Deploying the right tools
To develop products, the Biotec team collaborates with a number of medical and industry professionals, who represent valuable sources of experience, information and documentation. The company also partners with universities, especially their bio-engineering and the medical/surgery departments.
“We collect needs and requirements from all the partners, and they are recorded in one or more specification sheets and then transferred to our product development team,” says Piccoli. “These specifications enable us to analyze technical feasibility and provide the foundation for initial sketching and drafting on the PC (personal computer).
“Until a few years ago, we used 2D software, which was inadequate for developing assemblies; it didn’t allow is to take into account proper matching and interference among parts. The final output was a twodimension drawing, which had little practical use.”
In 2010, things changed with the adoption of 3D CAD, which sped up initial sketching and enabled Biotec to analyze the feasibility of a solution in detail.
“The values of technology and know-how are hot-stamped in our btk brand, and we can support such values only by selecting the best tools and partners,” says Peloso. “On one hand, we expand our knowledge through official partnerships with universities and key industry players; and on the other hand, we deploy tools that help our engineering and manufacturing staff develop and design specific products.
“It is essential for us to have a common tool in order to interact with all applications and processes – design, production and surgery.”
That’s where Solid Edge comes in.
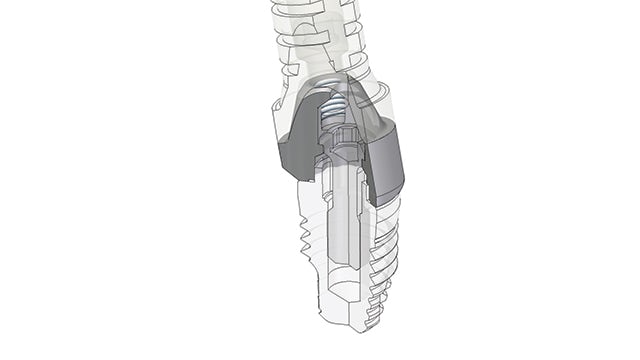
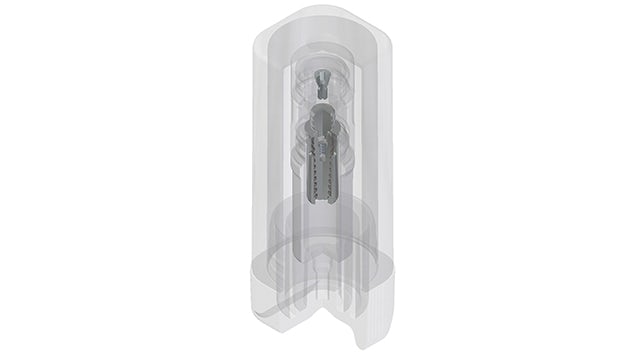

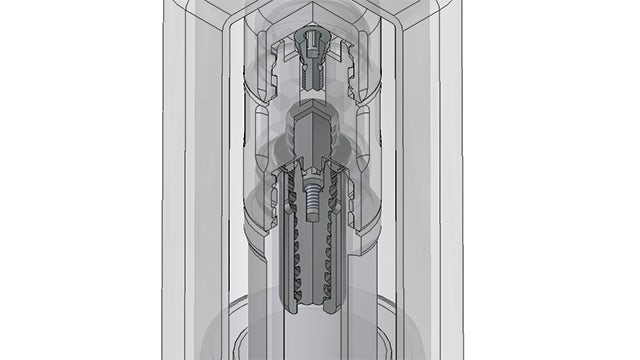
Marking a turning point
With Solid Edge, Biotec engineers can utilize a unified language inside and outside the company from early in the development process. The technical drawings, which require specific knowledge and expertise, have been replaced by the mathematics of 3D models. From such models, the 2D drawing is generated. The 3D model is the starting-point for all operations downstream.
“In the past, the 2D draft was handed out to the machine operator and converted into a machining program, with consequent issues in terms of cycle time and interpretation,” says Piccoli. “Now, with a unified and advanced technology source, we have eliminated all transitions, conversions or translations that caused an inevitable waste of time and input errors.”
The adoption to Solid Edge was a turning point for the company’s renovation and expansion project under the new btk brand. Before 3D was deployed, using solids-based design versus surface modeling, Biotec manufactured mechanical pieces rather than anatomic parts.
“Without an accurate digital model, we couldn’t achieve specific shapes and curves on machine tools,” says Piccoli. “Using Solid Edge, we can translate model geometry into machining programs through our CAM (computer-aided manufacturing) tools. With this approach, we have achieved part compliance, virtually eliminating idle time on the machine tools. As we machine one work piece, we prepare another program offline, which enables virtually uninterrupted production.”
Biotec found that with Solid Edge, the company is able to effectively collaborate with surgical teams, who now receive the implant geometry directly from the manufacturer to program and guide operations in the dentist’s office. This is possible because Solid Edge can handle a wide range of data formats, including Parasolid® software, IGES, STEP, DXF and DWG.
“The clinic provides the patient’s 3D model, which we integrate with our dental implant to simulate and plan the surgical operation,” notes Piccoli. “The 3D geometry is also used to prepare documentation, deliverables and the physical prototypes of our implants.” In this scenario, the synchronous technology of Solid Edge helps Biotec engineers suppress or simplify complex features or geometries very quickly, without having to rebuild the history of the model. With synchronous technology, users no longer have to choose between constraint-driven or history-free modeling, no longer have to be a programmer to re-use a model, and no longer need to worry about using data from multiple CAD systems.
“The benefits of Solid Edge with synchronous technology are amazing,” says Piccoli. “We also manufacture some mechanical equipment internally, and the use of synchronous technology delivers huge benefits when we design sheet metal for the support of parts to be submitted for heat treatment. We have created shapes that would be hard to imagine utilizing a traditional environment.”
Simcenter Femap serves as the starting point
To implement a full-featured design system, Biotec uses Solid Edge with Simcenter Femap™ software, finite element analysis (FEA) technology from Siemens Digital Industries Software. When designing very small parts like the pins of dental implants, an accurate analysis of stress and load distribution on the mechanical components and on the jaw bones is needed.
“Performing an analysis is mandatory, unless you are willing to spend a long time on mechanical tests, which would be difficult due to the tiny dimensions of our products,” notes Piccoli.
Today, Simcenter Femap is frequently the starting point for the design cycle at Biotec, not only for mechanical engineering, but also for the identification of the geometric shapes that guarantee the best fit for the patient’s anatomy.
“We try to collect as much information as possible to bring our simulation as close to the real thing as we can,” says Peloso. “Recently, we faced the challenge of having to make an ultra-short implant. Simcenter Femap played an essential role as short implants don’t seem to offer adequate load distribution at first sight. The simulation with Simcenter Femap was confirmed by physical tests and indicated that with accurate design and suitable geometries, even better load distribution can be achieved with few spirals.”
Part of the team
Biotec’s designers are supported by the Siemens Digital Industries Software partner Tech-Value, the value-added reseller (VAR) that worked with them throughout the implementation process.
“I have been in touch withTech-Value for eight years, including the period when I worked in a different company,” says Piccoli. “When we selected the design and development technology, we also considered service and support. Tech-Value has always been very supportive; whenever we have a problem, they respond to our needs and take action in real time.
“For instance, during the latest training course, we found some critical concerns that were resolved immediately, because we had established a very efficient input and feedback process that delivers quick and practical solutions. We think of the Tech-Value staff as part of the team, rather than suppliers.”
Using Solid Edge…we have achieved part compliance, virtually eliminating idle time on the machine tools.
Biotec